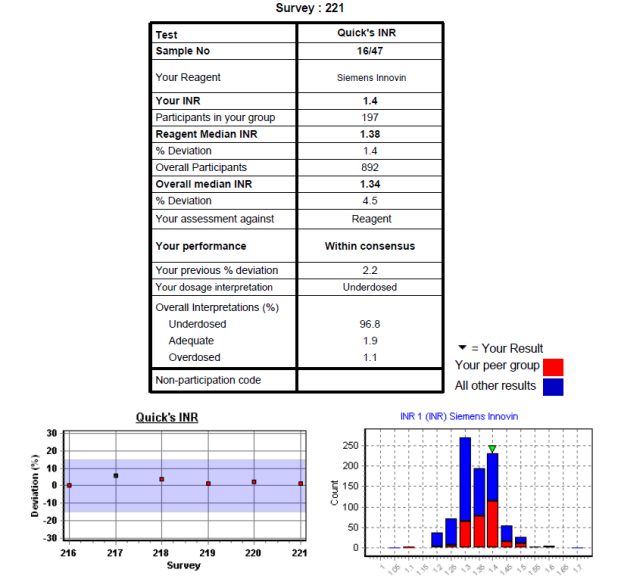
Data Analysis and Performance Assessment
The purpose of External Quality Assessment is to compare performance of individual laboratories or test centres with their peers. EQA is a retrospective form of quality control - participants do not know the target range or expected results for their test, unlike quality control.
The data analysis process is described briefly below - for a detailed explanation, please download below.
Adobe Acrobat document [39.8 KB]
Screening Tests (Level 1)
The median result for each sample distributes is taken as the target value. Peer groups are determined where applicable by reagent or method. A laboratory result within 15% of the target value is considered "within consensus" for INR, PT, APTT and clauss fibrinogen. A 20% limit is applied for heparin dosage assessment and thrombin times. Results outside of these limits are termed "outwith consensus".
Factor Assays (Level 2)
The median result for each sample distributes is taken as the target value. In most cases, the all method median is used as a target, the detailed guide above explains when peer groups are employed. A series of grades are determined based on the spread of results around the median (target value): the closest 50% of results are awarded an A grade, the next 20% a B grade, then C, D and Erespectively for results further from the median. Thus the 5% of highest and 5% of lowest results for an assay will be awarded an "E" grade. The combination of grades in successive surveys is then used to determine whether a participant is within or outwith consensus. For more detail, please download the file above.
For screening tests and assays, outwith consensus results based on 3 consecutive surveys are deemed "persistently outwith consensus". In such cases, a letter offering support will be sent to the participant from the Scheme Director.